Of Molecules and Minds: The Science of Ayahuasca
There’s a beauty and sense of wonder that comes from knowing how the world works, and the nature of the psychedelic experience dovetails almost perfectly with that awareness.

Psymposia is a 501(c)(3) nonprofit research and media organization that offers critical perspectives on drugs, politics, and culture. We rely on contributions from our readers and listeners. Your support is vital to sustaining Psymposia.
Support Psymposia’s independent journalism on Patreon and help us drive the Mystery Machine! We’re a bunch of meddling kids who are unmasking the latest shenanigans on the psychedelics beat.
Biochemistry is complicated. In this article I’ve attempted to distill one of the most complicated systems in the known Universe (human biology) down into an accessible 2,000 word piece. Consequently, I had to make some simplifications, deliberately tossing some details in order to communicate the larger patterns—the lock-and-key analogy is one example of this. If you want to learn more, I’ve included links to educational resources whenever possible.
AYAHUASCA: A MOLECULE EYE’S VIEW
As the psychedelic renaissance continues to build up steam and shake off a half-century of hibernation, the popular conception of drugs such as psilocybin and LSD is beginning to change. No longer are psychedelics just pharmacological cluster bombs, ready to push a hapless user to insanity. Instead, a new image, still in development, reveals these drugs as medicines for ailing souls.
No psychedelic drug has captured the popular imagination as a healing substance quite the way ayahuasca has. Celebrities from Lindsay Lohan to Sting have publicly endorsed its healing potential, and it has even wormed its way into popular television shows such as (believe it or not) Law and Order: SVU.
However, despite changing popular conceptions of psychedelics, particularly ayahuasca, the dialogue remains firmly planted in the domain of the therapeutic and the spiritual. While a quick Google search can return hundreds of articles, forum posts and trip reports that deal extensively with the psychological, therapeutic, and spiritual ramifications of experiences like those engendered by ayahuasca, what the discussion often lacks is a firm scientific footing—some context about how these drugs work on a chemical level, an explanation of what’s actually happening inside your body and brain when these drugs are consumed.
This feels like something of a missed opportunity: There’s a beauty and sense of wonder that comes from knowing how the world works, and the nature of the psychedelic experience dovetails almost perfectly with that awareness. There’s a profound symmetry between the wonder you feel learning how a biological or physical process happens and the wonder you feel when your mind is expanded by a psychedelic. Seeing a flower as a complex and emergent property of a hundred million chemical processes, and seeing that same flower as a beautiful manifestation of an interconnected, spiritual universe are not that different; they may even be the same.
DMT AND SEROTONIN
Ayahuasca is made by making a crude extraction from two very specific plants: Banisteriopsis caapi (which is the ayahuasca vine itself) and Psychotria viridis. While different curandero/as have their own recipes that can include various admixtures (and some leave out the P. viridis entirely), if you find yourself talking to an up-and-coming tech bro at Burning Man about his visionary ayahuasca experience, you’re almost certainly talking about the combination of those two plants for one very special reason: DMT. While ayahuasca without DMT can be a profound experience, it is the oral DMT that creates the visual fireworks, and its near-mythical status in the family of psychedelic drugs definitely adds to the mystique surrounding ayahuasca.
After boiling the plants for several hours, a few important molecules have migrated from the plants into the water. DMT, obviously, is one, although the others are arguably more important: harmine, harmaline, and tetrahydroharmine, which are all very similar molecules and belong to a family called beta-carbolines. It is these three molecules that make the ayahuasca experience possible. Ordinarily, DMT is not orally active, which means that if you were to sit down and take a pill of DMT, it would have no effect on your consciousness. While it has visionary effects when inhaled or injected, when eaten it is (normally) worthless.
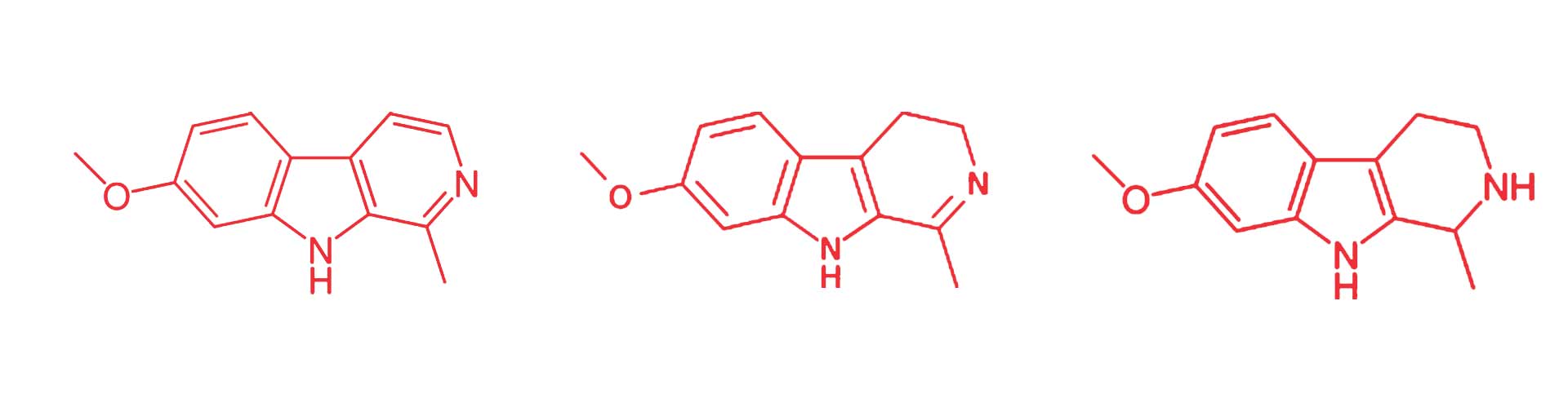
All of this is a quirk of biochemistry, which will be important later. Organic molecules, such as DMT, harmine, and our own neurotransmitters, gain their function from their shape: all of them are basically bundles of identical carbon atoms arranged into different configurations (with a handful of other atoms like Oxygen and Nitrogen thrown in). The shape of a molecule determines what its effect is; form determines function. At this level, biology and biochemistry are essentially applied geometry: The physical substrate of our body is largely carbon, no different than a diamond, or a charcoal briquette, but all that carbon in our bodies and brains happens to be organized into exquisite and dizzyingly complicated shapes which fit together in just the right way. From that intricate clockwork, life emerges.
When you look at the shape of DMT, you’ll notice it looks quite a bit like the shape of another well-known molecule: serotonin. As everyone knows (thanks to concerted advertising campaigns from the pharmaceutical giants) serotonin is a neurotransmitter, occurring naturally in the brain, helping to regulate the complicated dance of neural information processing. This symmetry means that in the body, DMT interacts with other molecules in much the same way that serotonin does: The classic metaphor is a lock and key. In this case, if serotonin is the key to a variety of different locks, DMT is a knock-off key similar enough that it can open some of the same doors. This is where its psychedelic effects come from. Before we get there though, we still need to determine why DMT isn’t ordinarily orally active, but is in ayahuasca.
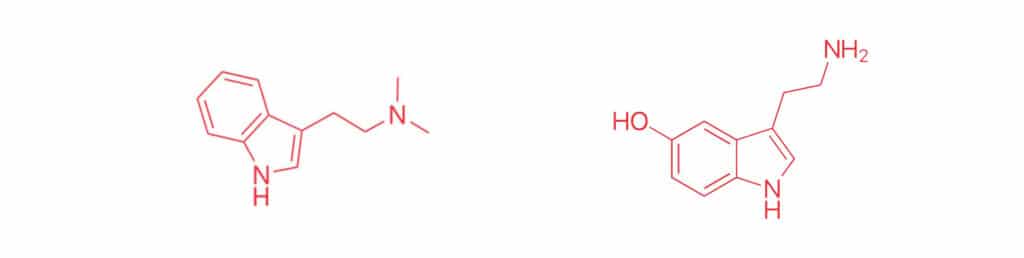
When serotonin is released in the brain and body, it serves a very particular purpose; once that purpose is filled, it’s in the interest of the body to get rid of it. This is accomplished through the use of enzymes, which are biochemical machines that facilitate chemical reactions that otherwise would be too slow, or wouldn’t occur at all. The enzyme we’re interested here is called L-monamine oxidase A (MAO-A). From its name, someone literate in biochemical jargon can figure out it takes monoamine molecules (like serotonin), and degrades them through a process called oxidation. Coming back to our lock-and-key metaphor, imagine a lock that fit a particular key—but rather than locking or unlocking a door, it was immediately snapped in half. In everyday bodily functioning, this is all well and good, all part of the delicate balance of metabolism. But for those looking to explore consciousness, DMT looks enough like serotonin that it too fits into our imaginary key-chewing machine. When DMT is eaten, it passes from the stomach into the bloodstream, where it goes directly to the liver and is immediately chewed up by MAO-A into something that has no interesting biological activity whatsoever.
This is where those three beta-carboline’s enter the story. When they bump into MAO enzymes, they bind to the same place that DMT and serotonin do, but rather than getting degraded, they just sit there, clogging up the machinery and rendering the enzyme at least temporarily useless. Imagine stuffing the lock of our key-chewing machine with gum. When DMT (which would ordinarily be degraded by MAO-A) enters the body at the same time as the beta-carbolines, enough of the MAO-A is compromised by the MAO-inhibitors that the DMT can make it through the gauntlet of first-pass metabolism and continue on to its final destination: the brain, where the magic really happens.
DMT IN THE BRAIN
Before we keep going, it’s worth mentioning that while the story of the DMT, MAO, and MAO-inhibitors is somewhat specific to DMT and Ayahuasca, what comes next is not. Other orally active psychedelics can survive first-pass metabolism on their own and make it to the brain largely unmolested. Once they get there, however, they all behave in largely the same way, DMT included.
Once the DMT has reached the brain, the fact that it is the same shape as serotonin once again becomes relevant. In the same way that its geometric similarity means it will get degraded by MAO-A, it also means it can activate the same switches in the brain that serotonin does, acting like a ‘pseudo’ neurotransmitter.
When a neuron wants to send a signal to the next one in its pathway, it does so by releasing neurotransmitters into the small gap (called a synapse) between them. The molecules diffuse across this gap until they run into their target neuron. The surface of the neuron is covered in little proteins called ‘receptors,’ which, upon meeting a molecule they recognize, briefly change their shape from the ‘off’ shape to the ‘on’ shape. The signal, which began with the release of neurotransmitters, is completed and the information has jumped the gap from one neuron to another. Turning a receptor ‘on’ can have a variety of different effects, including changing the likelihood that its neuron will fire, or altering the internal function.
When the DMT (which began its life in a plant in the Amazon jungle) finally makes it into the brain, it will flow into those synapses; because it looks a lot like serotonin, it will flip those receptors looking for serotonin into the ‘on’ position, even though no neuron released any serotonin. This is the basic mechanism for all psychedelic drugs; from LSD, to psilocybin, to weird things like 25i-NBOMe: They sneak into the synapses and ‘trick’ the serotonin receptors into activating, causing all kinds of changes in the functions of everything from individual neurons all the way up to the highest levels of consciousness.
So far, we’ve talked about serotonin receptors like they’re all mostly the same and broadly, that’s true (they’re all looking for serotonin, after all). But that doesn’t make them identical, and scientists have found enough differences to classify serotonin receptors into seven types, which are then broken down into 17 subtypes with a wide variety of functions. Most are pretty uninteresting to psychedelic scientists, but there are two that seem particularly important: serotonin-2A (5-HT2A), and serotonin-2C (5HT2C).
All psychedelic drugs activate the serotonin-2A receptor: some do it more than others, but it’s a requirement for psychedelic effects. Despite knowing that the serotonin-2A receptor is very important for the biology of psychedelics, scientists are still figuring out what its normal function in the brain is. We know it’s involved in regulating mood, appetite, sexual behavior, learning and memory, as well as some physical responses such as blood vessel constriction and temperature control. It is mostly found in a particular part of the cerebral cortex (known as Layer 5), where it appears on neurons that help communicate information from one brain region to another. Some researchers have speculated this is how psychedelics create their mind-bending effects: By altering how different brain regions talk to each other, the brain becomes chaotic and somehow, this creates the experience of a higher state of consciousness.
The serotonin-2C receptor is a little bit more mysterious. It is similar enough to the 2A receptor in that almost any drug that activates one will also affect the other, so it can be very hard to tell what effects come from the 2A receptor and which ones come from the 2C receptor. There is a lot of evidence that the serotonin-2C receptor plays a role in regulating mood and anxiety: Many of the common pharmaceutical treatments for depression and OCD affect activity at the 2C receptor, and we know that it helps regulate the release of dopamine (another key neurotransmitter) in brain regions important for learning and addiction. It has been suggested that the non-addictive nature, and anti-addictive power of drugs like LSD and psilocybin come from their activity at the 2C receptor, which alters function in addiction circuits.
Eventually, the ayahuasca experience ends. After four to six hours, even with the beta-carboline’s slowing things down, enough of the drugs are metabolized away that normal consciousness reasserts itself and we return to this world. Different metabolites may float around your system for a while, but eventually it all gets washed away.
A SOURCE OF WONDER
While many psychedelics seem to force contemplation of profound and often overwhelming thoughts and questions, ayahuasca, in particular, seems to invite a sort of wonder. It is a drug that can only exist because it interacts in precisely the right way with our own incredibly delicate biochemistry. DMT, harmine, harmaline, and THH exist in just the right shape and fit into our neurochemical clockwork in just the right way to produce profound outcomes. Even small changes to the shape of any one of those molecules might result in an inactive pile of nothing, or even a lethal toxin.
That sense of wonder is what psychedelic science should capture. Rather than reducing a magical experience down to dull, technical jargon about agonists, antagonists, Ki values and dose-response curves, it should enhance the awe we feel and show us that whatever wonder we feel at the beauty and glory of Creation, it is probably more wonderful still.
Hey! Before you go… Psymposia is a 501(c)(3) non-profit media organization that offers critical perspectives on drugs, politics, and culture. We strive to ask challenging questions, and we’re committed to independent reporting, critical analysis, and holding those who wield power accountable.
Our perspectives are informed by critical analysis of the systemic crises of capitalism that have directly contributed to the unmitigated growth of addiction, depression, suicide, and the unraveling of our social relations. The same economic elite and powerful corporate interests who have profited from causing these problems are now proposing “solutions”—solutions which both line their pockets and mask the necessity of structural change.
In order for us to keep unpacking these issues and informing our audience, we need your continuing support. You can sustain Psymposia by becoming a supporter for as little as $2 a month.
Thomas Varley
Thomas Varley has a BA in Neuroscience from Hampshire College, and studies Clinical Neuroscience at University of Cambridge.





